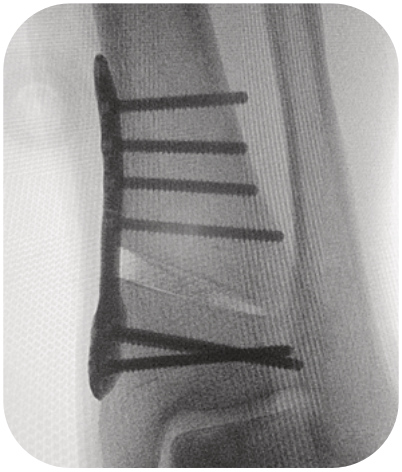
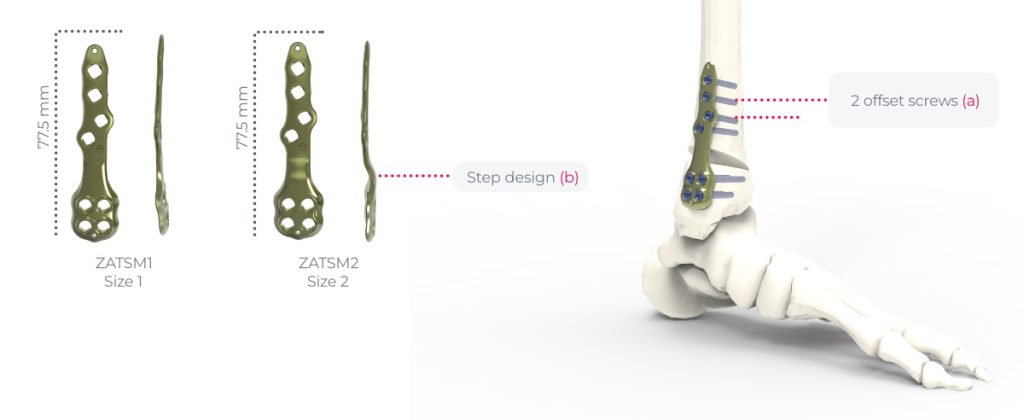
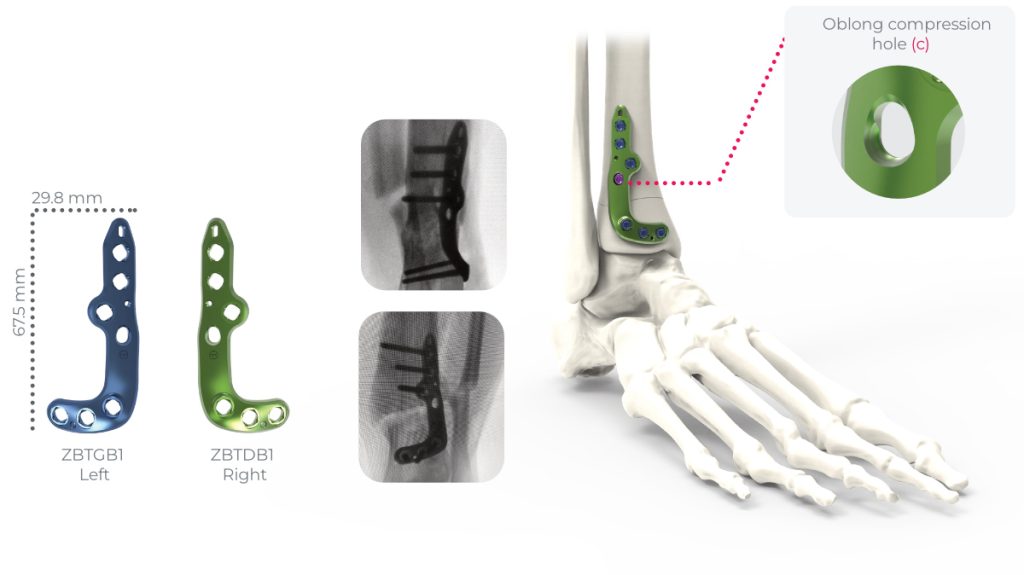
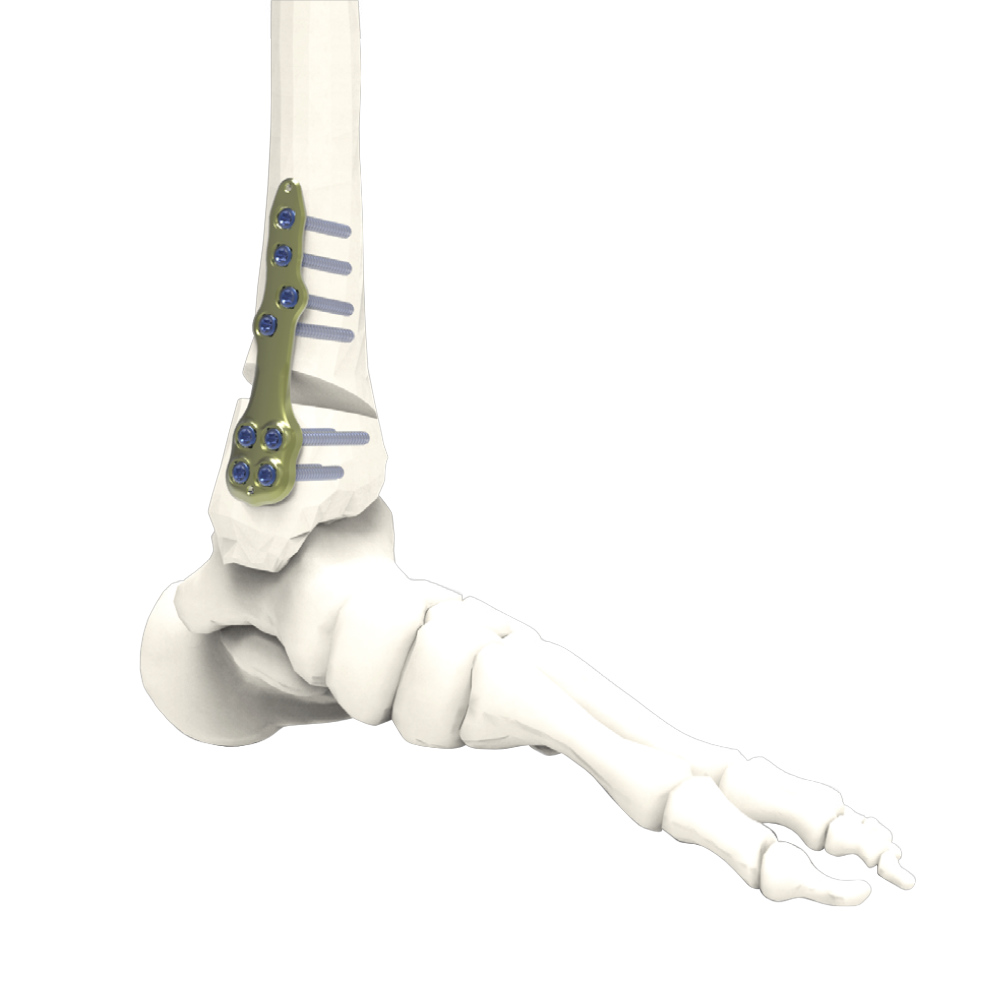
the implants of the Activmotion S DTO range are intended for bone reconstruction of the ankle joint in adults, including fixation of fractures and osteotomies of ankle, distal tibia and fibula.
2 offset screws for improving the mechanical features of the assembly (a).
Step design to optimize congruency of the plate according to the opening (b).